Oric Pharmaceuticals (ORIC)·Q4 2025 Earnings Summary
ORIC Beats Q4 as Clinical Data Strengthens Path to Phase 3
February 23, 2026 · by Fintool AI Agent

ORIC Pharmaceuticals delivered a strong Q4 2025, beating EPS estimates by 26% while reporting potential best-in-class clinical data for both lead programs. The stock rose 5.4% on the announcement as the company raised $264M from top-tier healthcare specialist funds and extended its cash runway into 2H 2028—well beyond the anticipated Phase 3 primary endpoint readout for rinzimetostat.
Did ORIC Beat Earnings?
Yes—ORIC beat EPS by 26%, reporting a loss of $0.30 per share versus the consensus estimate of approximately -$0.41.* The beat was driven by lower R&D spending as manufacturing costs for rinzimetostat declined, partially offset by higher personnel costs.
*Values retrieved from S&P Global
The R&D expense decline of $6.1M YoY reflects lower rinzimetostat drug manufacturing costs and reduced spending on discontinued programs, while G&A increased due to higher personnel costs and professional services.
What Changed From Last Quarter?
Three significant developments distinguish Q4 2025 from prior quarters:
1. Clinical Data Inflection for Rinzimetostat
The company completed Phase 1b dose exploration in prostate cancer and selected provisional RP2Ds for combination with AR inhibitors. Key efficacy signals:
- 55% PSA50 response rate (11/20 patients), with 40% confirmed
- 20% PSA90 response rate (4/20 patients), all confirmed
- 76% achieved >50% ctDNA reduction (13/17 patients)
- 59% achieved ctDNA clearance (10/17), exceeding standard of care benchmarks
2. Enozertinib Shows Compelling CNS Activity
Phase 1b data in NSCLC patients with EGFR exon 20 and PACC mutations demonstrated:
- 100% intracranial ORR in both EGFR exon 20 and PACC patients with measurable CNS disease
- 67% ORR (1L) in EGFR exon 20; 80% ORR (1L) in EGFR PACC
- Competitive safety profile with low treatment discontinuation rate
3. Capital Position Strengthened
Raised $264M from top-tier healthcare specialist funds through private placement and ATM programs, extending runway to 2H 2028.
How Strong Is ORIC's Cash Position?
ORIC ended Q4 2025 with $392.3M in cash, cash equivalents, and investments, up significantly from $274M at the end of 2024. With a proforma cash position of $412.3M (including a subsequent $20M ATM raise), the company has runway into 2H 2028—critically, beyond the anticipated primary endpoint readout for the Phase 3 rinzimetostat study.
The capital raise positions ORIC to execute multiple registrational trials without near-term financing overhang—a critical de-risking factor for clinical-stage biotech investors.
What Are the Key 2026 Catalysts?
ORIC outlined an aggressive catalyst calendar for 2026 across both lead programs:
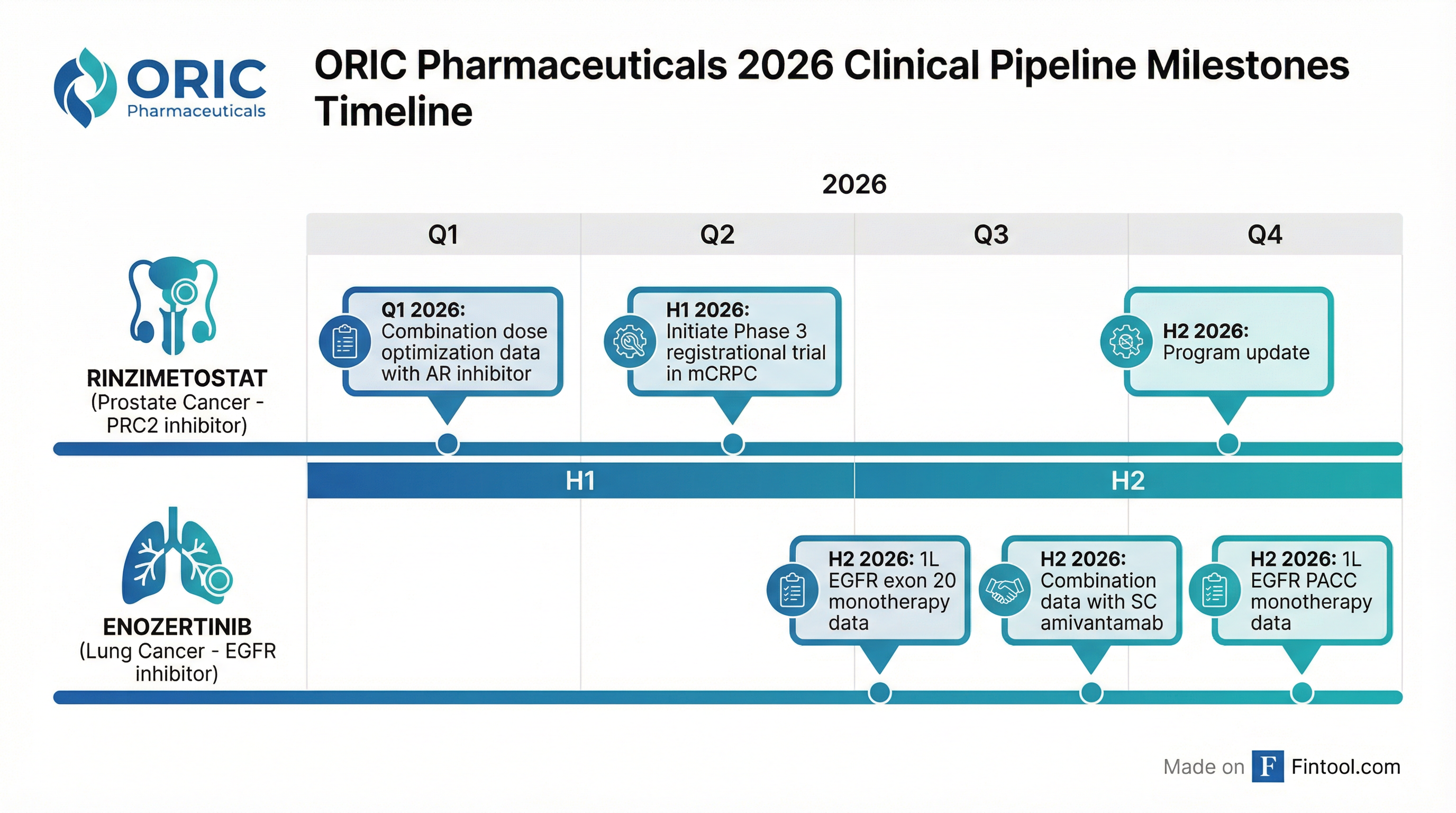
Rinzimetostat (Prostate Cancer)
Enozertinib (Lung Cancer)
How Did the Stock React?
ORIC shares rose +5.4% on earnings day, closing at $11.71 on February 23, 2026. The stock has rallied approximately 200% from its 52-week low of $3.90, reflecting improving clinical data and strengthened balance sheet.
The positive reaction reflects investor confidence in the clinical data package and extended runway, though shares remain ~22% below the 52-week high achieved earlier in the year.
What Did Management Say?
CEO Jacob M. Chacko, M.D. framed 2025 as a "transformational year" and emphasized the company's positioning for registrational advancement:
"2025 was a transformational year for ORIC highlighted by clinical data that further demonstrated the potential best-in-class profiles of rinzimetostat in prostate cancer and enozertinib in lung cancer. Those data, along with substantially extended cash runway, position us well for 2026 and beyond as we advance our programs towards registrational studies."
Key management claims to track:
- "Potential best-in-class" efficacy for both programs—validation will come from Phase 3 data
- Cash runway "into 2H 2028"—assumes current burn rate continues
- Multiple data readouts in 2026—execution risk on timelines
Full Year 2025 Summary
For the full year ended December 31, 2025:
The full-year net loss was essentially flat YoY despite advancing two programs into late-stage development, reflecting disciplined capital allocation as discontinued programs wound down.
Key Risks and Concerns
Clinical Execution Risk: Phase 3 trials carry binary risk. While Phase 1b data is encouraging, late-stage trials in oncology have historically high failure rates.
Competitive Landscape: Other PRC2 inhibitors are in development from competitors. ORIC must demonstrate differentiation in Phase 3 to capture market share.
Burn Rate: At ~$35M/quarter operating expenses, the company will consume significant cash through Phase 3 execution. Any trial delays could pressure the runway.
Regulatory Path: The FDA approval pathway for combination therapies in mCRPC remains complex, and ORIC will need to navigate biomarker selection and trial design carefully.
The Bottom Line
ORIC delivered a clean Q4 beat with the loss narrowing significantly YoY. More importantly, the company has de-risked its path to registrational trials with $264M in fresh capital and compelling Phase 1b data for both lead programs. The stock's +5.4% reaction suggests investors are increasingly pricing in Phase 3 optionality, though binary clinical risk remains. Key near-term focus: Q1 2026 dose optimization data for rinzimetostat ahead of Phase 3 initiation.
This analysis was generated by Fintool AI Agent using public SEC filings and market data. Not investment advice.